
In 1900, he explained, with Ernest Rutherford, that radioactivity is due to the transmutation of elements. Planck later called discrete units of energy as quanta.įrederick Soddy was an English radiochemist and monetary economist. In 1901 Max Planck, German physicist, proposed a theory that energy did not flow in a steady continuum, but was delivered in discrete packets. Marie Sklodowska Curie Marie Skłodowska-Curie, Polish physicist and chemist, discovered the radioactive elements polonium and radium. Thomson used a cathode ray tube for his experiment.Įrnest Rutherford, British chemist and physicist, studied radiations emitted from uranium and thorium and named them alpha and beta. Thomson discovered the electron in the year 1897. Wilhelm Roentgen, a German physicist, on 1895, produced and detected electromagnetic radiation in a wavelength range today known as X-rays.įrench physicist Antoine Henri Becquerel discovered radioactivity while working with uranium salts, which might emit penetrating X-ray-like radiation when illuminated by bright sunlight. 
Goldstein concluded that in addition to cathode rays, there is another ray that travels in the opposite direction. Goldstein, a German physicist discovered that tubes with a perforated cathode also emit a glow at the cathode end. In his investigations of the conduction of electricity in low pressure gases, he discovered that as the pressure was lowered, the negative electrode (cathode) appeared to emit cathode rays. He developed the Crookes tubes, investigating cathode rays. Sir William Crookes, was a British chemist and physicist worked on spectroscopy. He proposed that electricity was made of discrete negative particles and proposed the term electron to describe the fundamental unit of electrical charge. In 1873, he formulated classical electromagnetic theory and made quantitative connection between light and electromagnetism. James Clerk Maxwell was a Scottish theoretical physicist. In 1869, Dmitri Mendeleev created the first version of the periodic table of elementsof all known elements and predicted several new elements to complete the table. It was later shown that the glow was produced by cathode rays.ĭmitri Ivanovich Mendeleev was a Russian chemist and inventor. Plucker found that the electric discharge caused a fluorescent glow to form on the glass walls of the vacuum tube (cathode ray tube). This became the basis of theįaraday discovered that when the same amount of electricity is passed through different electrolytes, the mass of substance liberated/deposited at the electrodes is directly proportional to their equivalent weights.Įlectrolysis is a method of using a direct electric current (DC) to drive an otherwise non-spontaneous chemical reactionĮlectrolysis is the passage of a direct electric current through an ionic substance that is either molten or dissolved in a suitable solvent, resulting in chemical reactions at the electrodes and separation of materials. In 1832, Michael Faraday reported that the quantity of elements separated by passing an electric current through a molten or dissolved salt is proportional to the quantity of electric charge passed through the circuit. In chemical reactions, atoms are combined, separated, or rearranged. 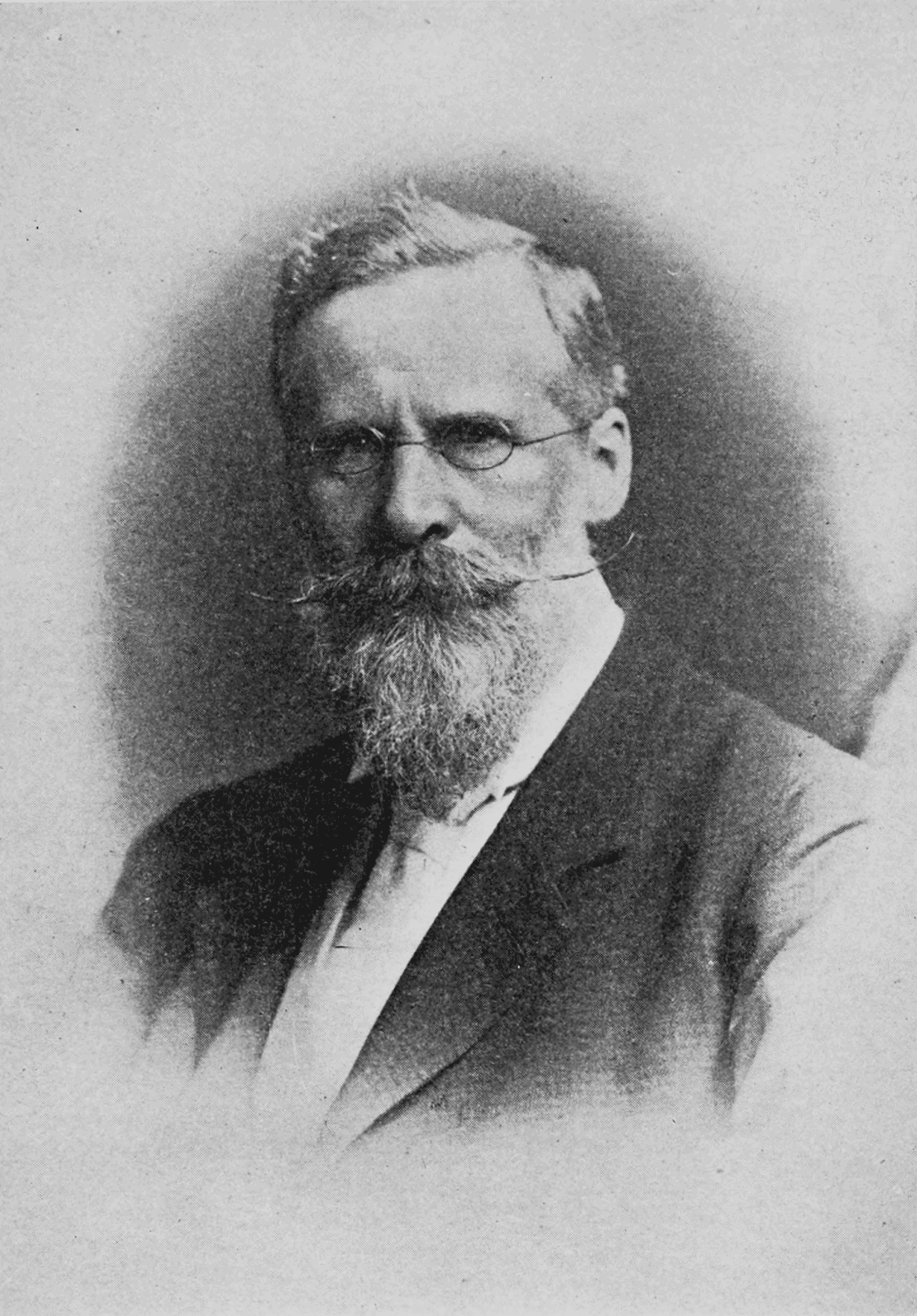
Atoms of different elements combine in simple whole-number ratios to form chemical compounds.Atoms cannot be subdivided, created, or destroyed.Atoms of a given element are identical in size, mass, and other properties atoms of different elements differ in size, mass, and other properties.Elements are made of extremely small particles called atoms.In 1803, John Dalton Proposed an "atomic theory".įive main points of Dalton's atomic theory: John Dalton was an English chemist, meteorologist and physicist. 
In 1704, Sir Isaac Newton, an English physicist and mathematician proposed a mechanical universe with small, solid masses in motion. He coined the term átomos, which means "uncuttable" or "the smallest indivisible particle of matter" in greek. Democritus (400 B.C.)Democritus proposed that matter was composed of tiny indivisible particles.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
